This application shows the rapid screening and quantitation (20 s per sample) of carcinogens (Aristolochic acid and its metabolite aristolactam) in medicinal plants using DPiMS™-8060.
Aristolochic acid, which is the target of this analysis, is found in plants of the genus Aristolochia, and is classified as a carcinogen, including their plant forms. This article introduces an internal standard method using naproxen for rapid screening and quantitative analysis of four types of aristolochic acid contained in Aristolochia debilis and Asiasarum sieboldii and aristolactam I, which is one of its metabolites, using the Shimadzu DPiMS-8060 direct probe ionization mass spectrometer kit.
Probe electrospray ionization method (PESI) or Direct Probe Ionization MS (DPiMS) is a unique interface technology from Shimadzu. Thetechnique successfully incorporated the best of both worlds; high ionization efficiency of ESI a nd simplicity of MALDI. This was achieved by using an ultrafine needle


Scanning Electron Microscopy image of a PESI probe used in DPiMS
DPiMS, with the PESI technology implemented, is characterized by the microscopic sampling volume and high sensitivity for the amount sampled. In a typical application, compounds at a low ng/mL concentration range can be readily detected by DPiMS, in which case the actual abundance of compound subjected to MS can be as small as a few attograms. For this, DPiMS can be a robust solution for the routine analysis of high-complexity matrices, such as plasma or food extract, as contamination of MS hardware can be perfectly mitigated. Moreover, direct sampling from solid samples causing minimal destruction might enable new applications such as real-time analysis of live tissues or cells.
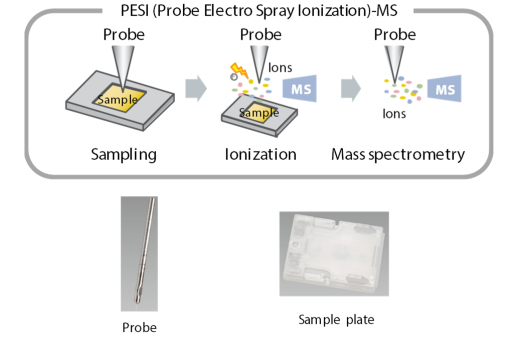
principle of probe electrospray ionization method the probe is thrust into the sample on the sample plate, and sample molecules are ionized by applying a voltage to the sample adhering to the probe surface.
Sample Preparation: The following two types of solutions were prepared for use in sample preparation.
- Methanol/water (70/30, v/v)
- Ethanol/water (60/40, v/v) : 2 mM ammonium formate, including 10 ppb naproxen (internal standard)
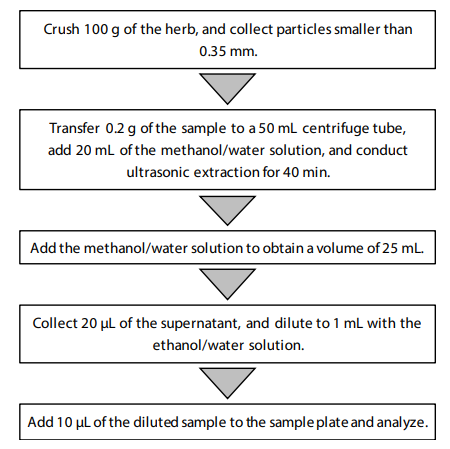
Workflow of Sample Preparation
Measurement Conditions:
- Sampling stop time : 50 ms
- Sampling position : -46.0 mm
- Ionization stop time : 220 ms
- Heat block temp. : 30 °C
- Interface voltage : 2.3 kV (+) / -3.0 kV (-)
- Probe cleaning time : 0.05 min (+) / 0.05 min (-)
- Probe cycle speed : 2.78 Hz
- DL temp. : 250 °C
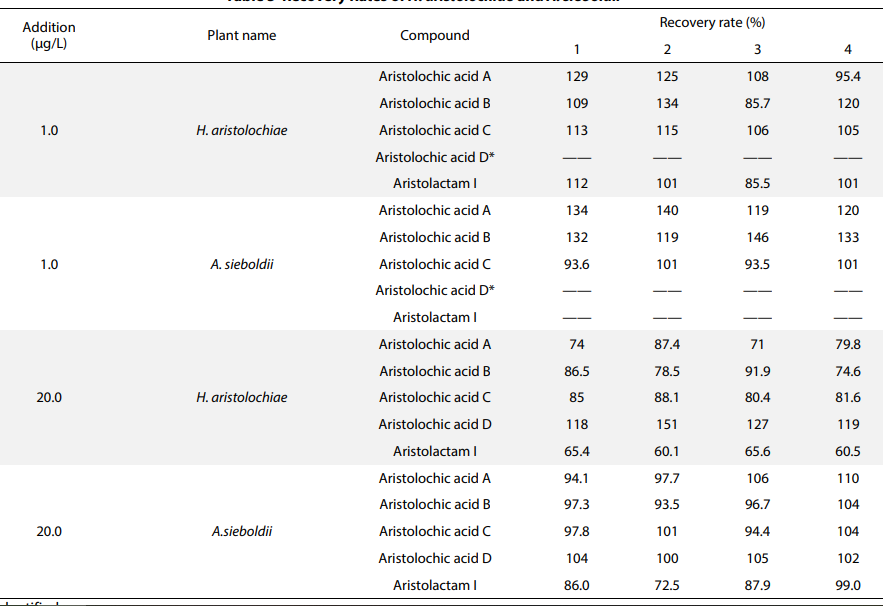
Recovery Rates of H. aristolochiae and A. sieboldii
- Life Science - Evaluation of Amyloid-β Aggregation by Fourier Transform Infrared Spectrophotometer (FTIR)
Instruments and Measurement Conditions: a Shimadzu IRTracer™-100 Fourier transform infrared spectrophotometer and a MicromATR™ attenuated total reflectance (ATR) measurement accessory (Czitek, LLC.) were used. The ATR method makes it possible to obtain a spectrum simply by placing the sample in close contact with the crystal, and cleaning after measurements is also simple in comparison with the transmission method. Moreover, the 9 reflection ATR crystal of the MicromATR accessory enables measurement with higher sensitivity than with the widely-used single reflection devices. Here, it may be noted that the optical system was purged with dry air because the peaks of the amide I band and water vapor overlap

Measurement Conditions
Experimental Procedure: Samples were prepared by dissolving amyloid-β (human, 1- 40) to approximately 10 μM with ultrapure water, and ATR measurements were carried out after allowing the samples to stand at 25 °C for 0.5, 1, 2, 3, 24, 48, or 120 h. In this experiment, 60 μL of the amyloid-β solution was dripped (30 μL × 2 times) on the ATR crystal, the water content was completely removed with dry air, and the exsiccated amyloid-β on the crystal was then measured
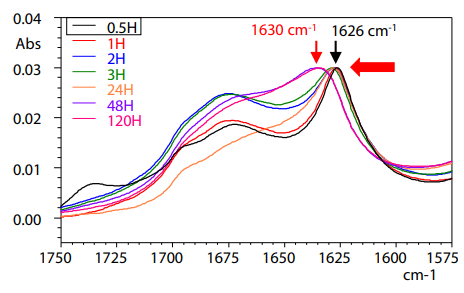
Amide I Band in Infrared Spectrum
The sums of the peak areas and the ratios of the high and low wavenumber regions of the β sheets (below table). The ratio after 0.5 h was 0.2836, suggesting a structure with a large fraction of antiparallel β sheets. After 120 h, the ratio decreased to 0.1860, clarifying the fact that conversion from the antiparallel β sheet structure to the parallel β sheet structure occurs over time, and the number of amyloid fibrils caused by formation of parallel βsheets also increases. This is the same result as the second derivative spectrum described above.

Sum of Peak Areas and Ratios of β Sheets
Amyloid-β aggregation was evaluated by using FTIR. The results of an analysis of the amide I bands in the infrared spectrum and second derivative spectrum suggested that a time-dependent change (conversion from antiparallel β sheet structure to parallel β sheet structure) occurs in the secondary structure accompanying aggregation of amyloid-β.